
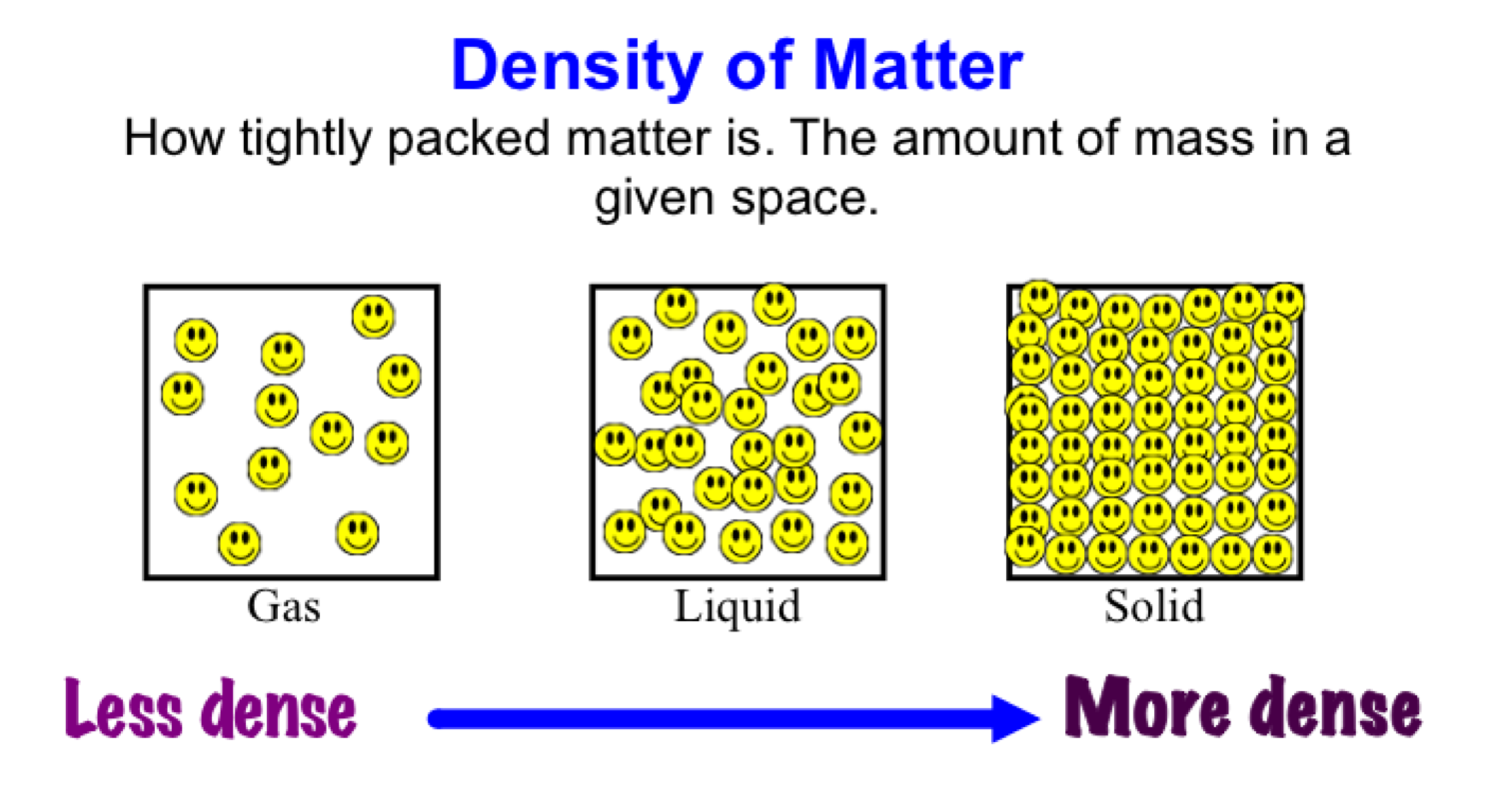
That's why the difference between heat of vaporization at 25C (energy required to break all H-bonds between 1 gram of initially slow moving molecules) and at 100C (energy to break all H-bonds of 1 gram of fast molecules) is LESS then the energy required to make all of those 1 gram of molecules faster. And as more molecules fly off, less energy is needed to break off the remaining bonds. It occurs more and more as you near towards it. Water’s lower density in its solid form is due to the way hydrogen bonds are oriented as it freezes. You don't need to wait until 100 degrees for vaporization to begin. This structure makes ice less dense than liquid water. 4 5 Much of an iceberg is below the waters surface, which led to the expression. 2 3 Smaller chunks of floating glacially derived ice are called 'growlers' or 'bergy bits'. As a result, when ice melts, it occupies less volume as water compared to. An iceberg is a piece of freshwater ice more than 15 m long 1 that has broken off a glacier or an ice shelf and is floating freely in open (salt) water. This multiplication factor accounts for the fact that ice has a lower density than water. A shallow convective overturn resumes until the lake is once more isothermal at 4 ☌. In the spring the surface water warms up and the ice melts. There are 3 different forms of water, or H 2 O: solid (ice), liquid (water), and gas (steam). By the time the surface water reaches 4 ☌ (39.2 ☏), the temperature of maximum density for fresh water, the density-driven convective overturn has reached the bottom of the lake. According to the formula, the water volume is calculated by multiplying the ice volume by 0.92. With 70 of our earth being ocean water and 65 of our bodies being water, it is hard to not be aware of how important it is in our lives.
Remember that when you apply energy to water, some of it will increase the avg kinetic energy of the molecules (related to the temperature) and some will be spent to break off all hydrogen bonding and send the molecules flying away (related to heat of vaporization at a GIVEN temperature). The formula used in this calculator is as follows: Water Volume Ice Volume 0.92. Water and ice has attracted much attention because of its anomalies pertaining to issues from galaxy to geology, astrophysics, biology, climate and to our daily lives 1,2,3,4,5,6,7.However, the. Its density is 0.98 g cm 3 at room temperature, as compared with only 0.92 g cm 3 for ice, a fact that must be explained by atomic/molecular theory.

How come only 40 calories can increase the water temperature by 75 degrees to its boiling point, if the specific heat property tells us that 40 calories can only increase it by 40 degrees?
